Best Online Casinos Ireland 🍀 Top Irish Casino Sites April 2024

All online casino sites in Ireland are very different from each other. And choosing the really the best online casino Ireland to play at can be difficult. GRDO (Gambling Rating Dashboard Online) monitors and generates a list of top casino Ireland for you in real time. With our expertise of 4+ years in the iGaming niche, we can objectively identify good online casino Ireland, make an honest list, based on the presence of bonuses, treatment of players, the authority of gambling brands, license and other important factors.
So, welcome to the GRDO panel! Choose an online casino you like, sign up and enjoy thousands of real money games at Ireland’s best products!
How the iGaming market is regulated in Ireland: a reminder for players
If we are honest about Irish casinos, the legislation here is rather vague: some of the gambling entertainment is under the scrutiny of the state. But there are no specific laws for online casinos. How it looks:
| Type of gambling | Regulation |
|---|---|
| Betting | Regulated ✅ |
| Casino | Unregulated ❌ |
| Online betting | Regulated ✅ |
| Online casino | Unregulated ❌ |
The current stage of evolution for casinos is the outdated and obsolete “Gaming and Lottery laws” of 1956. In those days, there were no online casinos in Ireland (or anywhere else, hah). So we can only turn to international licensing. Four licenses will be relevant for Irish gamblers:
- MGA (Malta Gaming Authority)
- UKGC (United Kingdom Gambling Commission)
- Gibraltar Gambling&Betting Association
- Curacao
If a player has chosen a licensed online casino in Ireland, he can be calm about his balance in the gaming club. All disputable situations (which sometimes happen) will be resolved in favor of the player. This is important: only adults are allowed to play at online casinos in Ireland. 18+
The current Emerald Isle government is discussing the regulation of gambling in the territory. At this stage, a full-fledged bill is under drafting.
Conclusion: lovers of gambling in Ireland do not face fines or other penalties for playing at real money online casinos. It is better to choose a project with a license MGA, UKGC, Gibraltar license, in the last case – Curacao. This way you will secure yourself and your money and enjoy the highest quality game. And the mission of GRDO — to help you make the right choice!
Top online casino Ireland: GRDO’s pick
If you are registering for the first time and want to do it right, follow the instructions after our ranking. And in front of you — the best casinos in Ireland on all parameters: from bonuses for new players to wager, loyalty systems and convenient ways to deposit.
How to create an account at online casino in Ireland
Firstly, you need to choose the gaming club that suits you best. We will tell you how to do it correctly in the sections below. You can safely rely on GRDO recommendations, as we ourselves have been playing in casinos for more than 3 years.
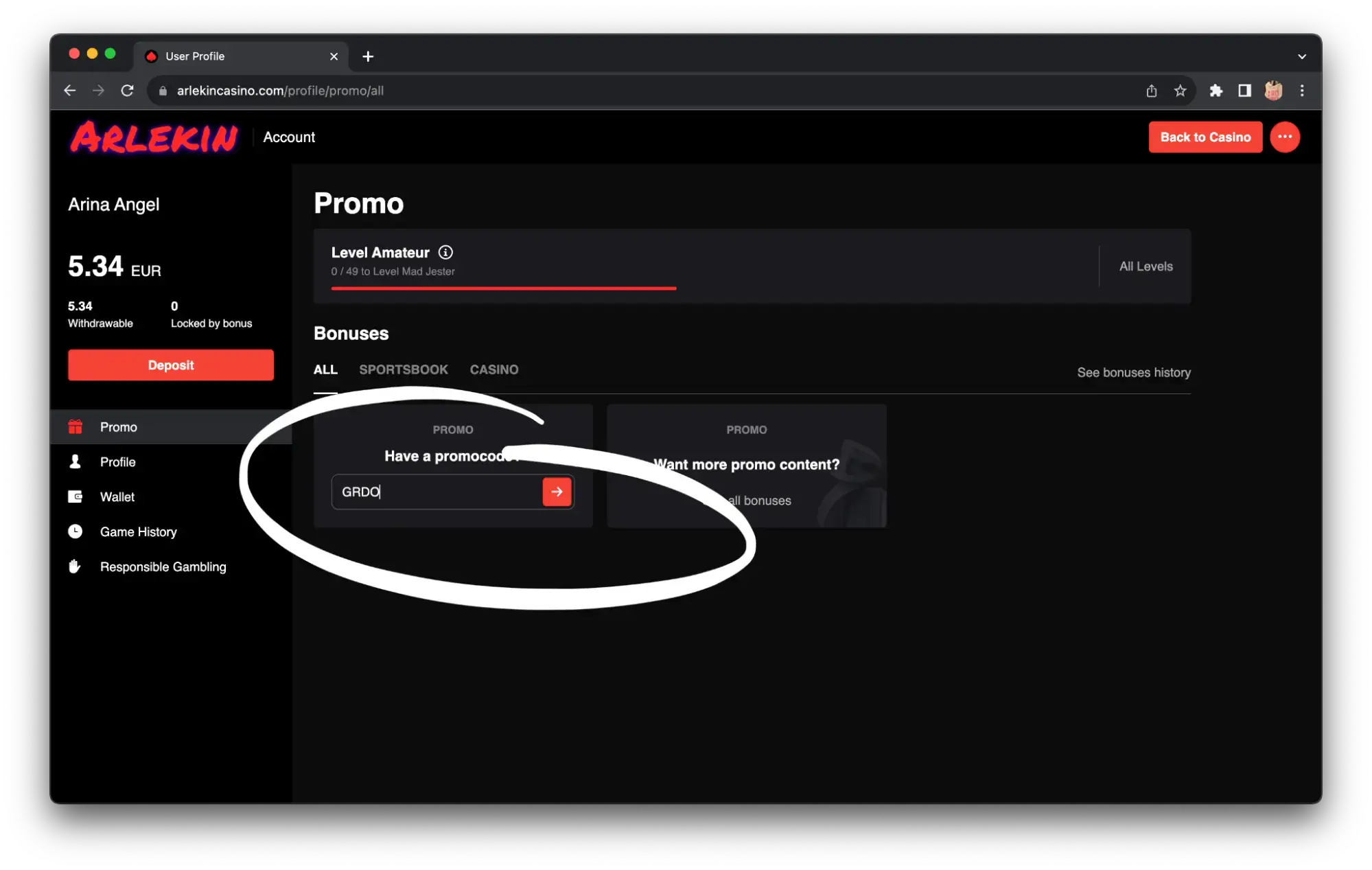
For those who are new to gambling, we will break down the registration process using Irish online casino Arlekin as an example. Step one — register in the project, you will need mail, phone number, login and password. If the registration form has a field for a promo code, be sure to enter the code GRDO – it will give a unique bonus in almost every Irish online casino.
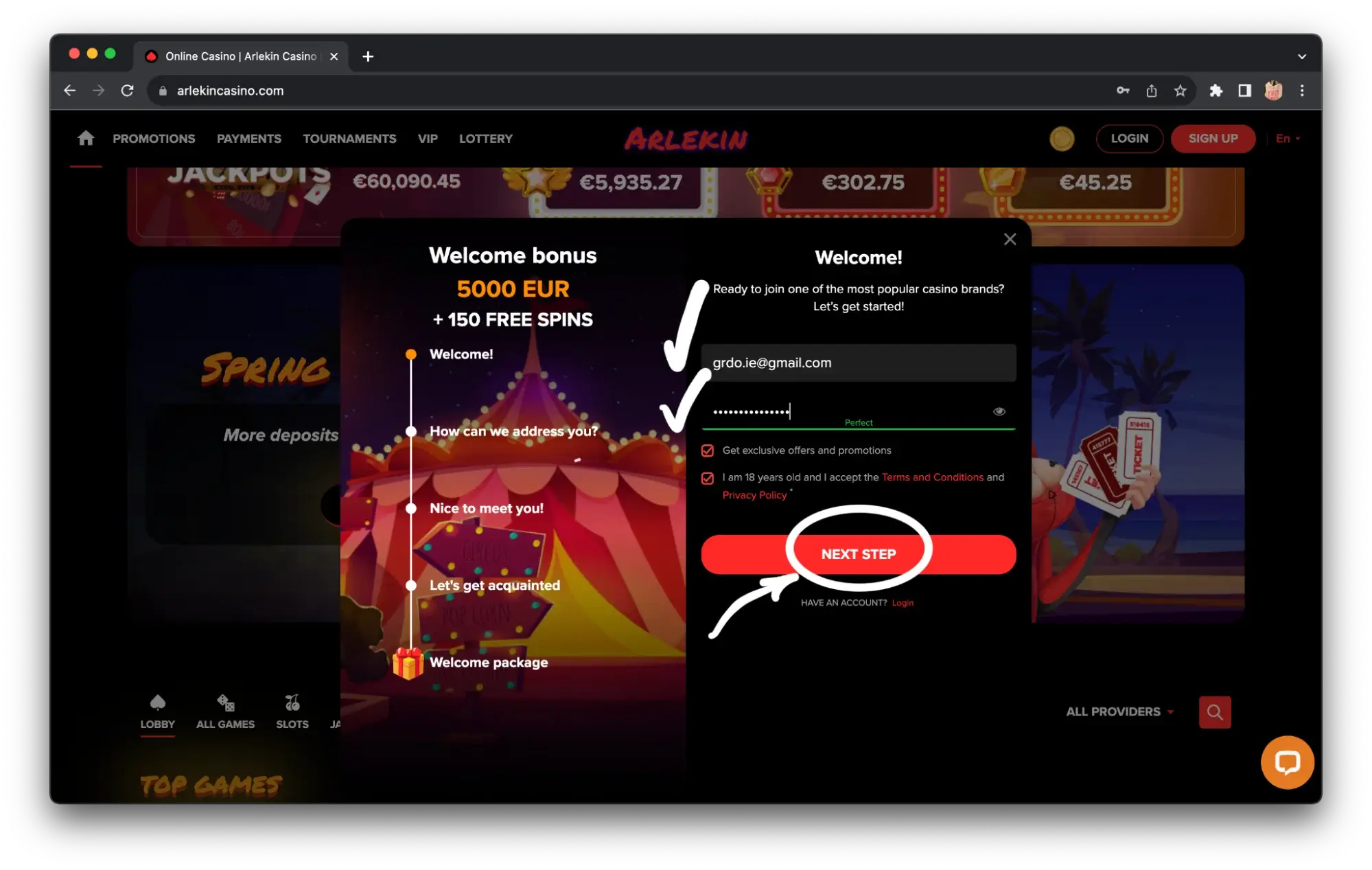
Step 2 — confirm your email address and phone number via SMS before making a deposit. In some projects, the promo code is entered already after registration in the personal cabinet.
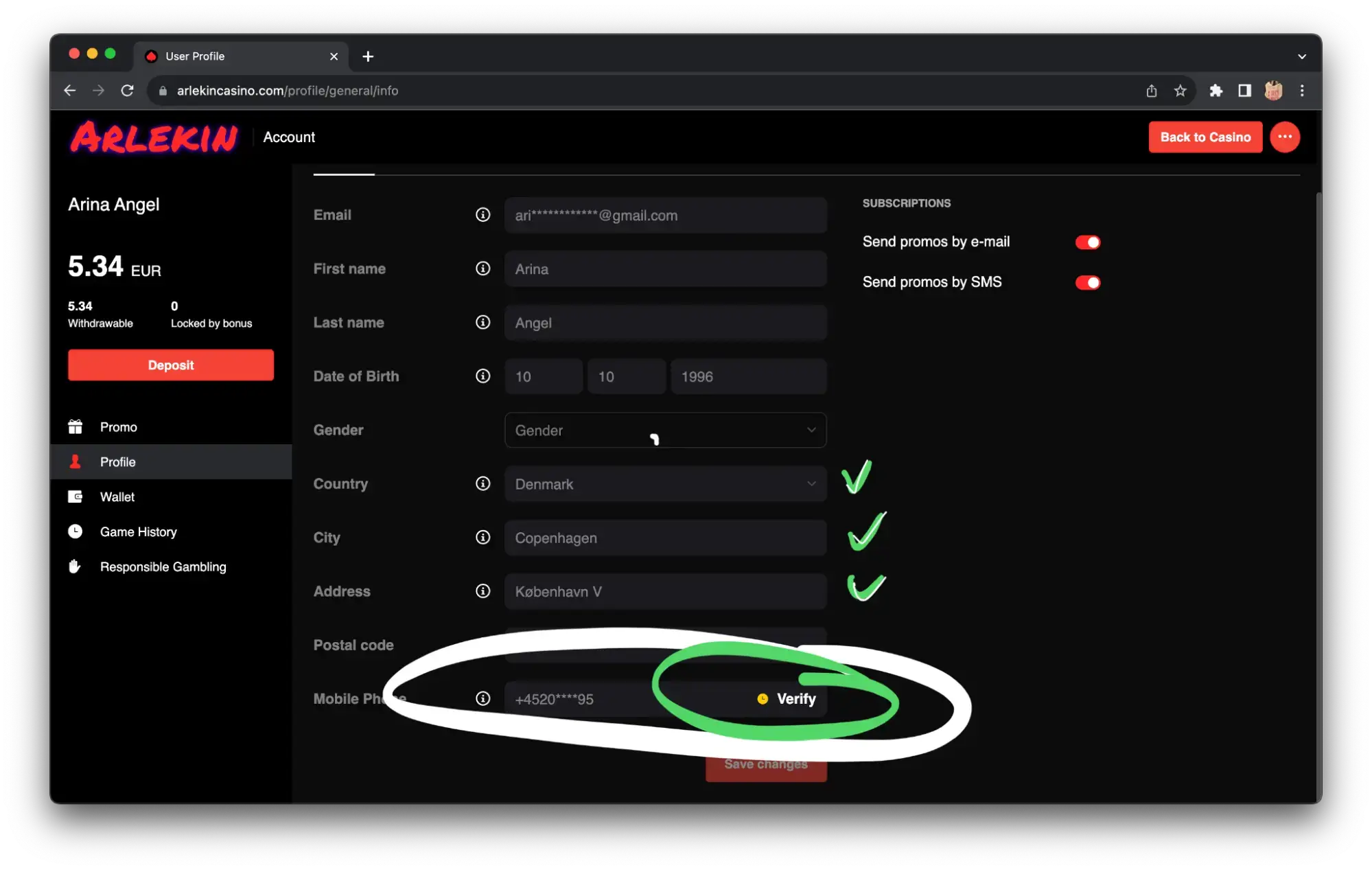
Step 3. Only after verification of your account via email and phone, you can make your first deposit and get high on first-class online slots and big winnings. And if you enter the promo code “GRDO”, you’ll get another nice bonus in addition to the welcome package.
Best online casinos in Ireland: how to make a deposit
After the registration procedure and account verification, it’s time to fund your gaming account! There are many options for funding your casino account in Ireland. The deposit can be made:
- From a bank card
- With cryptocurrency
- Through popular payment services
According to statistics, the most popular payment methods are debit and credit cards, cryptocurrency, Neteller and other e-wallets. In the rating of casinos from GRDO these deposit methods work in any of the presented projects. If an online casino does not have these deposit methods, we do not include it in our listing.
To replenish the account, go to the “Deposit” tab, choose the deposit method that is convenient for you:
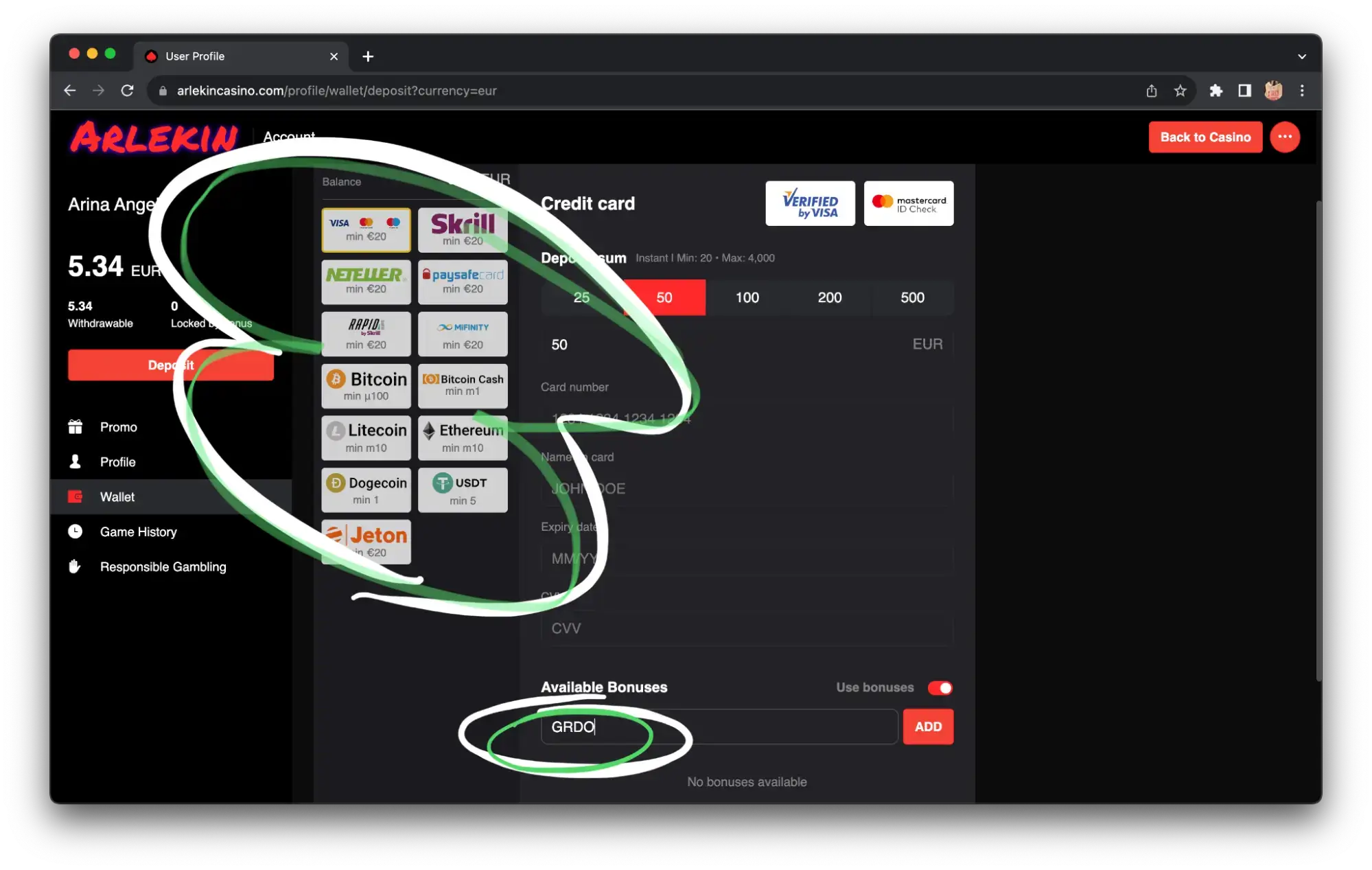
Enter the payment data and replenish the account. Also, sometimes you can enter a bonus code in the deposit tab.
The most popular payment methods on Ireland’s online casino sites
Let’s list the most easy and fast methods of funding your casino gaming account. Irish players prefer:
- VISA and MasterCard debit and credit cards — is probably the most convenient way. Cards are always at hand in your wallet, or registered in Apple Pay or Google Pay.
- USDT or USDC – ways to deposit cryptocurrency using TRC-20 or ERC-20 protocols. If you don’t have a wallet yet, it’s quite easy to start one in the “Wallet” app.
- Bank Transfer – casinos support such transfers with the note that the minimum deposit when depositing this way is much higher than usual.
- Jeton — a convenient app for casino deposits, currency exchange and keeping different currencies in one wallet. There is a handy app for Android users.
- Revolut — another e-wallet from which it will be convenient to fund your online casino account.
- PayPal is a rarer method at the moment, which is not available on most casino brands. Popular e-wallet.
- PaySafeCard is a popular method of depositing casino vouchers with a 16-digit code.
- Pay by Phone, Apple Pay and Google Pay are fast and convenient but rare methods, as officially organizations like Apple and Google do not make such payments directly to online casinos. Most often a player has to make a transfer to another individual, in return the casino adds money to the gambler’s account.
- Poku is a mobile transaction service without the need to link your bank card. Not available on most brands.
- Bitcoin or Ethereum are the most popular crypto ways to fund your account.
- Neteller is an old and convenient wallet for handling electronic money. Most of the Irish online casino brands are funded through it.
- Litecoin, Bitcoin Cash, Dogecoin. Many casinos are based on the SoftSwiss platform: this aggregator allows to quickly assemble the technical part of the casino project, and direct the team’s efforts to the visual part, media promotion. Basically, all SoftSwiss products include the ability to fund the casino account DogeCoins from Ilon Musk. So, if you suddenly staked or bought them for the sake of interest – here is a great way to spend Doge!
- Skrill and Rapid by Skrill is another payment system that makes players’ lives easier.
Zimpler, Trustly, EcoPayz and others — these wallets are also very convenient, but are more common in the USA and among players from Europe. But if anything, keep them in mind! These ways will be enough to keep your account empty. In addition, the casino will give you a generous first deposit bonus!
It is important: some bonuses are given when you deposit a certain amount. Carefully look at the terms and conditions of bonuses to increase your chances of winning at online casinos. Our rating shows the current bonuses and all the information on how to get them after registering on the project. And once again we remind you: most Irish casinos online have a bonus code GRDO.
Our promo code gives all new players a unique bonus: no-deposit free spins, % to the first deposit or other game benefits. Use it with pleasure!
How online Irish casino sites get on the GRDO panel
If we imagine there is a recipe for a good Ireland casino, it would look something like this:
- Big bonuses at the start.
- Constant promotions and extra bonuses for regular players
- A passable wager and convenient min dep
- Max bet size in online casino
- Professional tech support
- Wide selection of live games, slots from different providers
- Unique difference from other products
- No RTP manipulations
- VIP programs for high rollers
- Presence of tournaments and promotions, brand media presence
- Popularity among streamers
- And some extras 🍀
But we look even deeper: we study the players’ reviews, check whether they are inflated or not. Also, we study how recognizable and popular the casino brand is in the country.
And the main point is that the GRDO editorial team made a deposit with their own hands and tried the product we offer you on our panel. We may not write perfectly as journalists, but we definitely think like players.
If you are experiencing the iGaming industry for the first time, it is important for you to know everything in detail. So let’s go through each item from our recipe in more detail.
Welcome bonuses in Ireland online casinos
The basis of our recipe is a successful start in a casino project. Often, gambling establishments double or even triple a player’s first deposit. This is a good trend: your chances of winning grow right at the start.
However, there is a better option: a ladder of bonuses for 2—5 deposits. In this case, the online casino offers a long-term relationship between the platform and the player. Be sure to take this into account when choosing a project on GRDO. In that way, you would not change projects every week.
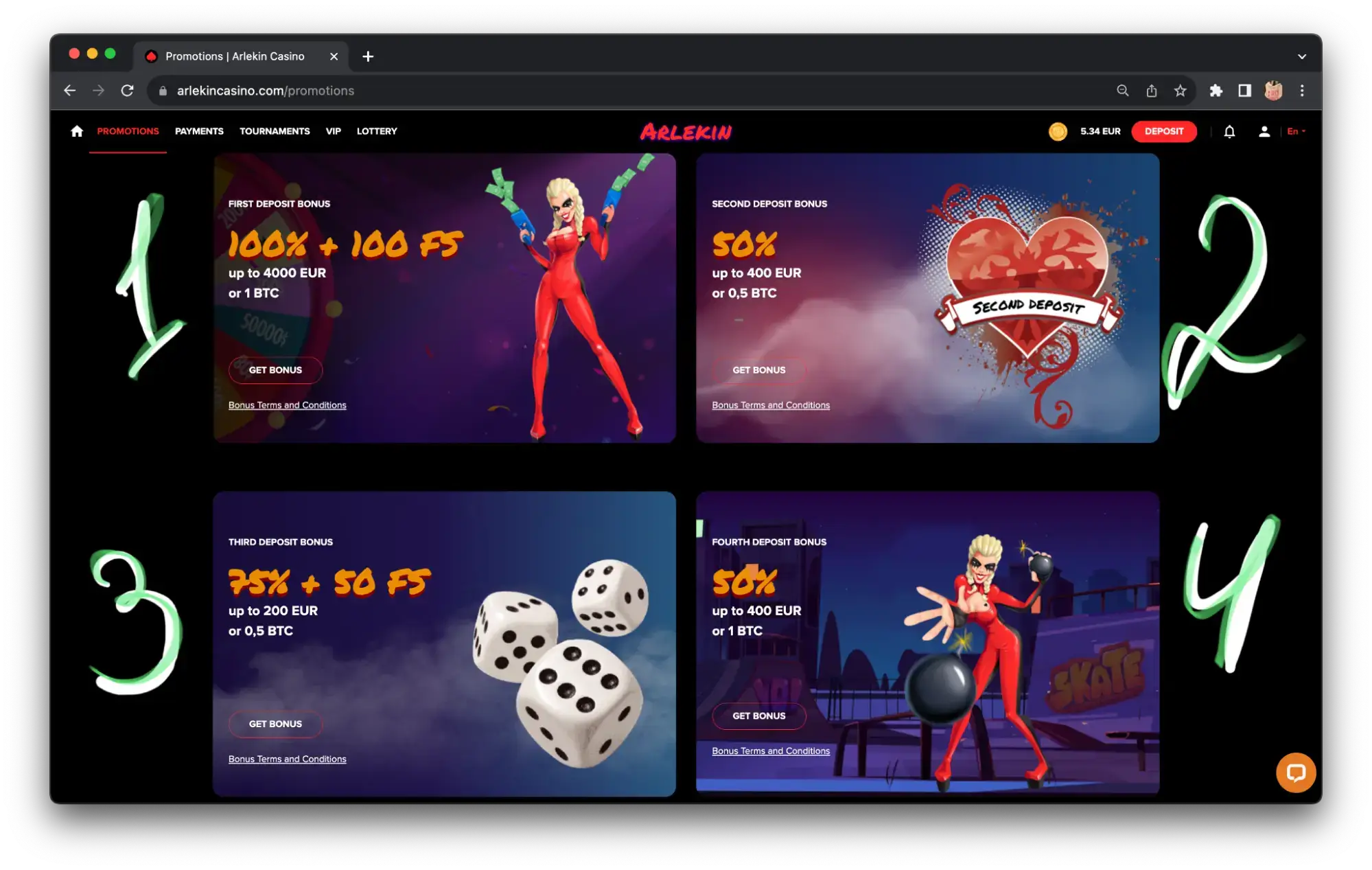
Reload bonuses & promo for regular players
All goodies for the newbies, but what about us, hardcore gamblers? For them, top casino Ireland offers weekly promotions and sometimes other benefits.
Most often it is a bonus to deposit on a certain day of the week, loot box or free spins on the weekend, as well as event promotions. But there are some projects that do better than others. For example, at Arlekin Casino, regular players rack up “achievements” – these are cards that appear during the game. They are given out for 5,000, 50,000 and 50,000,000 spins and other activities within the online casino.
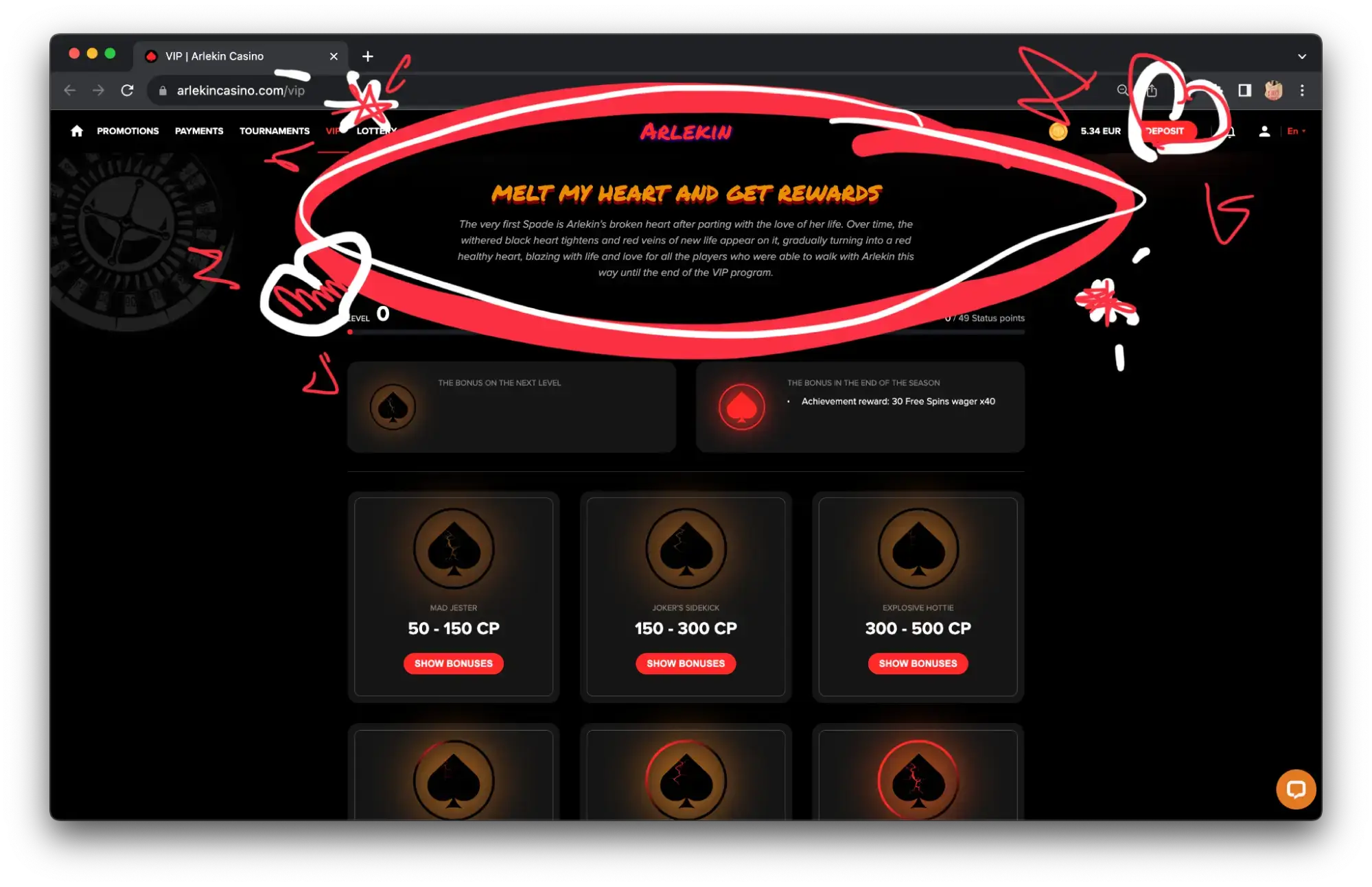
The cards look cool, the collection is available in the personal cabinet. In addition, each card gives the player a personalized bonus in the form of free spins, % to recharge and other in-game values. This is a unique bonus for Arlekin Casino players only. Other projects may offer different or similar bonuses.
Wager in online casino
The wager is the coefficient that is assigned to the bonus balance. A simple example: a player deposits 100 EUR and receives additional 100 EUR on the bonus balance. In order for this money to move from “bonus” to real money, online casinos introduce a wager — for example, x40.
Thus, the player must bet on the amount of the bonus multiplied by 40, in our case it is 4000 EUR. After that, the won money will turn into real money and can be withdrawn from the casino account.
Each casino and sometimes individual types of bonuses have their own wager. The lower it is, the better for the player:
- x3-x20 — good, low wager
- x20-x45 — average, normal wager. Such is present on most online casinos in Ireland
- x45 and above — high, bad wager. Most likely, during the wagering period, the gambler will simply lose money due to the long distance. If we in GRDO see such a wager at the product, we immediately underestimate it in the rating of the casino.
You can find the wager information in each brand’s card on our dashboard.
Amount of minimum deposit
Players often look at the minimum deposit: there is even a caste of gamblers who look for casinos with a minimum deposit of €1, €2, €3, €5. We don’t recommend this in general: you’ll have enough money for 10-20 spins, which in most cases will bring nothing. Conversely, the larger the deposit, the more varied your tactics become.
But usually the min dep is higher – on average in Ireland and other English-speaking countries it is from 10-20 to 40 EUR. For us, this is another opportunity to segment online casinos: the lower the minimum deposit, the more convenient it is for the player, which means that the brand stands in the rating above the rest.
Maxbet size
Beginning players are most often interested in the size of the minimum deposit, but for rich high rollers such a point as maxbet in online casinos is important. Many people are not interested in betting 10-20 euros: they want to play slots with a bet of 1000, 2000 euros per spin or more.
And surprisingly, not every online casino can offer a good max bet. Most often it is limited to 1000, maximum – 2000 euros per spin or bet. But there are also projects that allow you to make spins with a price of 4, 5, 10 thousand euros. Such online casinos get extra points in our rating.
Online casinos tech support
The customer support at the casinos is pretty easy to appreciate:
- Speaks in the local language without an interpreter;
- Available 24/7 by mail, messenger or online chat right on the casino website;
- Respond to a message within 1 minute;
- Excellent product knowledge;
- Treats every type of player with respect: beginners and high rollers alike.
Great variety of slots & live games
It is always difficult for beginners to choose a slot: there are so many of them in every online casino. However, this is only at first glance: in fact, there are not so many games with modern graphics, high RTP and interesting gameplay. As well as providers that produce cool entertainment.
With that in mind, here’s what we look at when scoring the best online casino sites in Ireland:
- A large selection of providers from the top 100 developers best online casino games Ireland
- On brand you can play any type of games: slots, live games, crush games, poker, etc.
- New slots appear in the casino on the day of release.
No RTP manipulations
In addition to a large selection of games, it is important that online casinos in Ireland do not lower the RTP artificially. Brands can do so even while being within a regulated market and on a license basis. If an online casino does not lower the RTP for players, it stands above the rest in our ranking.
VIP Online casino Program
An inclusive VIP program for regular players and those who can afford to play for large sums of money is a must at every good online casino. Now, VIP-programs are most often divided into levels, creating gamification for users or achievement systems.
All this keeps the project interesting and shows the online casino’s interest and good attitude towards its players.
Free online casino tournaments and promotions
There are a numerous popular tournaments that are organized by providers, i.e. game developers. For example, “Dream&Drops” by Pragmatic Play, which was later adopted by other providers such as Relax Gaming.
But in fact, the casino can independently hold various tournaments and promotions. And if a brand engages in such and encourages its players, it gets an additional point in our expert rating. For example, on the eve of 2024, CryptoBoss online casino created an Advent calendar, in which every day there were new bonuses for regular players: free spins, low wager bonuses, increased cashback and much more. In parallel, the online casino drew 3 cars.
It may seem that such promotions are exclusively for people with a lot of money, who are not sorry to spend real money in the casino, for example, several tens of thousands of dollars. But this is not the case. Going back to CryptoBoss, they have developed a mechanic where players are categorized:
- In the 1st category are high rollers who bet from 5–10 dollars and higher.
- In the 2nd category are mid-level players, whose bet size is from 2 to 5 dollars.
- In the 3rd category are beginner gamblers and those people who are carefully studying online casinos and make small bets from 0.2 to 1 dollar.
Each category has a winner who takes away an expensive car. And the winners who ranked from 2nd to 30th place in each group get impressive cash prizes.
Thus, online casinos increase interest in their project and give more opportunities to win to their players. And you, dear gamblers, should not overlook such promotions. In our ranking of the best online casinos in Ireland at GRDO, almost every brand has such promotions: from weekly competitions to live casino poker tournaments, including on mobile devices.
Extras: level of popularity among casino streamers
We closely monitor the activity of online casino brands in the streamer community. If popular media personalities often play in specific projects, it definitely increases their credibility. It means that the brand is not afraid to be public, does not cheat players, and withdraws winnings on schedule.
Ireland online casino sites review: 3 best options according to GRDO
So, it’s time to introduce you to the three best options to play online casino games for real money. These casinos are personally tested by us, have the greatest selection of games, top-notch support, elaborate levels of VIP-program and loyalty. This is where the most high-profile casino tournaments with huge prizes take place, it’s easy to deposit your game balance and also easy to withdraw your winnings.
Here come our representatives!
Arlekin Casino
A cyberpunk-style project with a charismatic mascot: the brash and daring girl Arlie, based on the prototype of Harley Quinn. Arlekin Casino is a licensed product of Curaçao with a wide range of games, fast withdrawal of the wins and no need to verify the account. You can do this later.
The chip of the online casino is the unique achievement system. In your personal account you can collect a whole set of Arlie cards, for each of them the player receives a bonus: free spins, percentage to deposit, no-deposit bonuses and much more.
- Licence: Curaçao
- Number of games: 5000+
- Minimum Deposit: 1-20 EUR depending on the payment method
- Withdrawal: from 5 minutes to 24 hours
- Bonuses: welcome, reload, special social bonuses, weekly and monthly bonuses, cashback, VIP bonuses, seasonal bonuses
- VIP-program: available
Kakadu Casino
Another terrific brand for gambling enthusiasts from the Emerald Isle. Kakadu Casino is a nominee of numerous awards. In 2023, the product received the “Best Bonus of the Year” award according to the prestigious AffPapa portal.
And the reward was received for a reason: in total Kakadu can award players up to 10000 EUR bonuses for 3 consecutive deposits and add to them a couple of hundred free spins. Bird of paradise flies on the wings of luck!
In 2024, Kakadu Casino reorganized their VIP program: it is now divided into 48 levels. Together with the casino’s mascot – the mischievous Kakadu bird – the player follows this path to the main treasure. What exactly — you will find out after registration.
- Licence: MGA
- Number of games: 4000+
- Minimum Deposit: 1-20 EUR depending on the payment method
- Withdrawal: from 5 minutes to 48 hours
- Bonuses: welcome, reload, special social bonuses, weekly and monthly bonuses, cashback, VIP bonuses, seasonal bonuses, lootboxes
- VIP-program: available, absolutely new and you can try it first 🙂
Abo Casino
The trendy Australian brand Abo Casino boasts the largest range of games of all kinds: live casino, poker, blackjack, scratch cards, crush games, roulette and many more. Luckily for us, the project works in several countries at once: Australia, Austria, Denmark, Great Britain and Ireland. So you can appreciate it at its best too!
Another great advantage of Abo Casino — absolutely incredible cashback for players: if you comply with the conditions of the permanent promotion on the casino site, the gambler can receive up to 20% cashback every day. For example, in most online casinos, cashback is awarded once a week or once a month.
- Licence: Curacao
- Number of games: 4000+
- Minimum Deposit: 1-20 EUR depending on the payment method
- Withdrawal: from 5 minutes to 48 hours
- Bonuses: welcome, reload, special social bonuses, weekly and monthly bonuses, cashback, VIP bonuses, seasonal bonuses
- VIP-program: available
Best Mobile Casino & App in Ireland
The trends of the last few years are obvious: more and more people are playing casinos on mobile devices or tablets (we approve of this, pretty nice experience!), using online casino apps, and even watching streams from their phones.
It’s a normal practice: I, for example, like to play slots on a long trip or – the coolest thing – chill with my tablet in the bath. The screen is still big, but it feels completely different from the MacBook. And in the last 2–3 years, a lot of people have switched to mobile devices in classic gaming, and in gambling in particular.
Casino brands, slot developers and providers are also feeling this trend. That’s why today there are as many as three variations for comfortable mobile casino play:
- Adapting the casino website to fit mobile screens.
- Creating an unofficial online casino application for Android and iPhone.
- HTML-5 technology, which provides a smooth and easy game on smartphones.
Mobile versions of online casinos
Until now, many online casinos do not have a mobile-friendly version. This is due to the fact that the development of adaptations takes time and requires enough resources. We at GRDO have selected for you the most convenient casino in terms of adaptation for any mobile devices with Android or iOS.

Casino Apps
You won’t find official real money online casino apps in Ireland. This is the policy of popular stores such as Google Play and AppStore. There should be no gambling entertainment inside.
Therefore, programmers on the casino staff create alternatives that are all just as player-friendly. With Android, everything is simple: an APK app is created, it can be downloaded on the casino’s official website or on its social networks.
Caution: do not download the application from unverified and unreliable sources! It may contain a malicious virus or steal your login and password in the casino. After that, you will lose access to your account and your money.
With the App Store things are a bit more complicated: more often than not, the app is disguised as a light-hearted game or a service for tracking body metrics, for example. When the user launches the app, a full-fledged casino site opens inside. It works from the application window, even if the casino domain is banned in the selected country. This technology is called Webview and is used in regions where casinos are banned and blocked by the local regulator.
HTML-5 for gamblers
HTML-5 is the coding language in which all new slots and games, and sometimes the functionality of entire websites, are written. It is a lightweight, user-friendly architecture that does not overload your smartphone and allows you to enjoy smooth spins in online casinos with high FPS.
Simply put, the mobile phone doesn’t heat up, the image doesn’t glitch or shake, games load quickly and don’t freeze even on the slowest devices.
Irish online casino bonuses: what they are and how they differ
Whichever online casino you choose to play at, each of them is sure to be full of bonuses and special offers for players. How not to get confused in this variety – we explain below.
The best Irish online casino games: what to play
Another factor of differences between online casinos that we are obliged to mention is the gamble assortment. In 2024, we can conditionally divide all casino entertainment into several categories:
- Live dealer casino games
- Slots developed by providers
- Crash games with instant winnings.
- Interactive games: adaptations of slots with a live dealer, loot boxes, scratch cards and instant lotteries, Bingo games.
Let’s talk about each of them in a little more detail.
Live dealer casino games
Sessions with live dealers are streamed online. The player feels as if he is in a real casino, but sees the game table, dealer, cards and chips on the screen. Some may think that this is not the same atmosphere, and perhaps it is really true.
But at the same time, live dealer games remain the most popular category of online casino in Ireland. At any table you may be joined by other players, and there is a live chat room for convenience and communication with the live dealer during the session. The dealer reads it and responds to comments from the gamblers at the table. Many players even make friends at online casinos!
There are quite a few games with live dealers. We will highlight the timeless classics.
BlackJack Live
One of the oldest games in the casino arsenal. The gameplay is simple: the dealer deals the cards, and the player must total 21 points or as close to it as possible. At the same time, the dealer plays against the players: if the gambler collected 19, and the dealer 20, the second takes everything.
Blackjack is characterized by the speed of sessions and extra chips that have been introduced in online casinos: you can make extra bets on the fallout of the same cards or get an extra win for 23 points. There is also classic blackjack and speed blackjack: in it, the dealer works faster, and the session time is significantly reduced.
Baccarat Live
In Baccarat, you are challenged to guess the outcome of a game between a nominal player and a banker. The dealer hands 2 cards each to the player and the banker, the winner is the one who gets 9 points or closest to this value.
Live Baccarat has many additional rules: for example, if one side scores less than 5 points, it gets a third card; if one side scores more than 10 points, they are discarded (e.g. 13 points automatically equates to 3).
Online casino players can bet on the victory of the player, the victory of the banker, a draw, the drop of two identical cards on either side. Most often the casino offers classic baccarat, speed baccarat and VIP baccarat for those who like to make big bets.
Roulette live
A canonical table game that has gained a lot of legends, memes, incredible success stories and crushing failures over the course of its existence.
The rules are simple: the player must guess where the ball will stop: you can bet on the color of the winning field (black or red), an even or odd number, a specific number or a range, for example, from 1-12. Pure luck!
That said, online roulette has additional modifiers and features that land-based casinos don’t have. These can be multipliers on certain winning numbers (this works in lighting roulette) or additional rules for different types of roulette.

Poker Live
Poker and its variants are very much appreciated by players at online casinos in Ireland, as the selection of these games is small compared to other entertainment. Yes, by typing “Live Poker rooms” into the search box, it will bring up more than 10 lobbies, but in fact they differ little from each other.
The main types of poker games in online casinos are Texas Hold’em, 2-card and 3-card poker and other varieties like Caribbean Poker or Oasis Poker. Tastes of poker players are specific, so it is recommended to play different modes and understand what will be interesting to you personally.
Slots online
Online slots are not just games; they represent a whole entertainment industry! There are numerous providers, each with their own vision of gameplay, additional features, and the ability to purchase bonuses, among other exciting characteristics. You can find all this and more in the special section on our website.
Slot games vary in terms of volatility levels. Some games offer evenly distributed wins on each spin or bonus round. While the maximum win in these games is lower and occurs less frequently, even a regular spin can reward players with a sum that’s x50-100 of their initial bet. These games are produced by companies like Pragmatic Play.
On the other hand, there are games where spins often result in no wins or minimal returns, well below standard odds. But if luck is on the player’s side, the winnings can be substantial, for example, 500 times the base bet. These are high and extremely high volatility games; many providers label them with a special symbol. The maximum win in these games is much higher, ranging from x20,000 to x150,000 from the base bet. Such games are regularly produced by Wazdan Gaming, Nolimit City.
Slots are a great modern entertainment. It is a must to try.
Crash games
Crash games such as Aviator, JetX, SpaceX and others are not widely available on the Emerald Isle. These games have become popular recently, but many online casinos have already put them in a separate section on the site.
Crash games are simple: there is an airplane flying on the screen, a rocket, the main character on a jetpack – visual adaptations vary depending on the provider. While the object is flying, the win multiplier grows. The player makes a bet before the flight starts, and can take the winnings at any time.
It sounds complicated, but in fact the mechanics are very simple and understandable to any category of players: from beginner gamblers to high rollers, for whom the crash game added the ability to make double or auto-bets, chat with other players and flight statistics.
For simplicity and the ability to quickly increase the budget, crash games are loved in many countries around the world. And Ireland is no exception.
Other interactive games
If all of the above categories aren’t enough for you, we have a few more types of entertainment found on Ireland’s online casino sites!
- Firstly, gaming projects have long since introduced classic games such as bingo, keno and other table games.
- Secondly, many popular slots such as Sweet Bonanza, Big Bass Splash have been adapted into a live dealer game show – and you owe it to yourself to try this as well.
- Third, online casinos haven’t skimped on modern entertainment: loot boxes from CS:GO and casual games, Monopoly, Plinko and even instant lotteries.
All of this is available at the online casinos in Ireland that you see on the GRDO website. We have specially selected the best projects so that all you have to do is to go and start the game.
The best game providers in Ireland online casinos
The production of games, organization of broadcasts with live dealer games are engaged in whole companies. In general, iGaming is a huge business, in which we, GRDO editorial staff, have been working for more than 3 years. In this section we will mention the most popular among Irish players and their games. If these companies are represented in online casinos, the project gets additional points in our rating.
NetEnt
Everyone knows NetEnt for its legendary titles: StarBurst and Gonzo’s Quest. In fact, the company has 200 amazing games in its portfolio. Netent has been on the market for more than 25 years and has established itself as a reliable provider and developer.
Pragmatic Play
Pragmatic Play needs no introduction. The industry giant has been producing 2 to 5 games since 2015. During this time they have made a huge number of global hits: for example, Sweet Bonanza, Gates of Olympus, Dog House. These games are online casino classics and many players start their journey with them.
Pragmatic pays a lot of attention to player protection and responsible gambling, developing the direction of live dealer games.
MicroGaming
Microgaming is one of the oldest companies in the market. They have been developing games since 1994, both for online and land-based casinos. The provider’s portfolio includes slots, live roulette, andar bahar and baccarat and even a couple of legendary games such as PlayBoy.
Hacksaw gaming
Hacksaw gaming is the least known provider on our list. But this makes it no less interesting. The company focuses on three gaming areas: online casino slots, scratch cards, and instant win games. Since 2018, the provider has gathered tens of thousands of fans in more than 20 countries.
Evolution Gaming
Unlike all the previously listed software providers, Evolution specializes only in games with live dealers. The company is present in literally every online casino, covering players’ requests for blackjack, roulette, baccarat and other popular games.
Online Casino & Gambling News Ireland
Fresh news from the world of online casinos, issues of regulation, responsible gaming, payments, stories of big wins and other interesting topics you will find in a special section with casino news on the GRDO website.
Land-based casinos in Ireland: a choice of popular places to visit
For those who want to experience gambling emotions in a land-based casino, we have prepared a selection of places in Dublin and other Irish cities, a must-see for every self-respecting gambler.
PlayLand Casino, Dublin
A finalist for BEST CASINO CLUB 2012-2014, the year-tested venue is ready to welcome players from all over the country. Strictly 21+.
🕐 Open: 10am to 1am
🏠 Address: 9 Talbot Street #8, Dublin
☎️ Contact: +353 (1) 874 83 33
D1 Casino Club
The best land-based casino in Dublin according to TripAdvisor with a huge number of positive reviews, in-casino events, free drinks and snacks for regular visitors.
🕐 Open: 10am to 5am
🏠 Address: 63 Lower Dorset Street St. Ignatius Rd
☎️ Contact: +353 (1) 830 67 64
The Fitzwilliam Casino & Card Club
Another Dublin gambling club. It has been operating for several years, tournaments are constantly held on the basis of the casino, the emphasis is on card games: poker, blackjack and other entertainment for gamblers.
🏠 Address: Clifton Hall, Lower Fitzwilliam Street
☎️ Contact: +353 (1) 611 46 77
Fitzpatrick’s Casino
The most beautiful casino in Dun Laoghaire, with a huge number of slot machines and a wide range of entertainment. If you’ve dreamed of visiting casinos that are shown in movies, this is your chance.
🕐 Open: 9am to 4am
🏠 Address: 94 George’s Street Lower
☎️ Contact: +353 (1) 214 4015
Responsible gambling at Irish online casinos
It is important to realize that it is up to both the online casino and the player himself to follow responsible gambling measures. Below you will find recommendations that will help you enjoy the game and not lose too much.
How to check if the casino complies with the measures on responsible gambling
Find out if there is an option to block your casino account through tech support. Players who are facing difficulties or gambling addiction (ludo mania) can ask to have their account blocked themselves. This is important: most casinos that care about responsible gaming will block an account once and for all. And since you specify personal data in the profile, you can’t return to the project.
Check if the casino has periodic notifications about how much time you have already spent in the game. Now not only casinos, but also some providers, integrate directly into their slots such reminders. It might look like this: “You’ve already spent 59 minutes in the game and lost 20% of your budget. Are you sure you want to continue?”. Such pooches bring overly gambling players to their senses and help them stop.
A good online casino prohibits access to gambling for people under the age of 18 or 21, depending on the jurisdiction. When KYC is passed, responsible gambling casinos instantly block underage players and prevent them from funding their accounts.
The project, which takes measures to protect players from gambling addiction, always has access to game history and loss statistics. These figures help you come to your senses and not lose your mind.
It is important to recognize that it is up to both the online casino and the player to follow responsible gambling measures. Below you will find recommendations that will help you enjoy the game and not lose too much.
How to behave as a responsible gambling player
Play only with really available money, which is not a pity to lose. Never take a risk by playing with your last money, on credit or on the pledge of property.
Never try to win back what you have lost by repeated deposits or increasing your bets in the hope that you will get lucky. This will only increase the amount you lose.
Set strict limits: this applies to the time of the gaming session and budget. Gambling is very addictive, so it is important to stop in time, take regular breaks to reduce the level of heat. If you schedule your monthly budget in an app or plan your spending on note, you can make a separate category of spending on “gambling entertainment”.
Remember that casinos are just that, entertainment, not a way to make money. Do not look for “100% win strategies” and do not believe that some person has cheated the casino, even if it is written in the media. This is all native advertising and an extremely ugly attempt to attract players to the project.
Avoid alcohol, drugs or medications that affect brain function while playing at an online casino. It is mandatory to be in an exceptionally alert state and soberly assess the risks throughout the game.
Contact specialized international and local organizations if you need help, use resources to help people with gambling addiction that are available on the Internet. Be sure to learn responsible gambling practices described by specialized organizations.
Remember: casinos are just entertainment, not a way to make money. Do not look for “100% win strategies” and do not believe that some person has cheated the casino, even if it is written in the media. This is all native advertising and an hideous attempt to attract players to the project.How tech support works in online casinos
Most often, the support in online casinos is divided into two groups:
- Support for all players. Handles all incoming requests
- VIP-support. Personal managers of players who bet huge amounts of real money and requires more attention to themselves
Support in online casinos helps to solve problematic issues, advises players, solves emerging difficulties with deposit or withdrawal, speeds up many processes. For example:
- Support can greatly speed up the process of passing KYC: it usually takes up to 48 hours to check your documents, but if you write to the manager, the documents will be checked almost instantly (we use this fly hack on every new casino project!).
- The support helps to replenish the account manually, if for some reason one of the payment methods stopped working. This is one of the most frequent problems with which players apply.
- The support service can solve the issue of bonus accrual. For example, you registered at one of the online casinos presented on our site, but forgot to enter the promo code GRDO. In this case, the player can create a ticket with a request, and the bonus will be credited post facto.
The main thing is that the player and the service employee understand each other. Therefore, in a good Irish online casino support should speak the language of players, even better – to be a native speaker of this language. Agree, the difference between someone who studied English at university or spoke it from childhood is huge. It’s even partially visible in our texts, hah!
Casino tech support is your best friend in the gambling world. Don’t forget, and feel free to create tickets, start chat rooms. On most Ireland online casino sites, the chat with tech support is in the bottom right corner of the screen.
The casino is scamming! What to do? GRDO Helpdesk
We know that gambling business is far from classic and it is quite difficult to get justice here. And in this respect, the GRDO panel is 100% on the side of players.
Notice: some of the sites on our list pay us affiliate fees for player registrations. This fact does not affect the position of the project casino in our listing in any way. Firstly – the facts, and the reward is just a nice bonus, thanks to which the project can develop and become the best rating of online casinos in Ireland.
Unfortunately, we cannot guarantee that every casino project on the GRDO panel will properly fulfill its responsibilities to players throughout its existence. Judge for yourself: the project may change management, one particular employee may behave incorrectly, as well as in online casinos often technical hiccups and difficulties.
Therefore, if you have a negative experience with one of the projects presented on the site, write to us in the feedback form or in the comments under the casino review. It can be anything: delayed payment of winnings, boorish attitude, difficulties with depositing or withdrawing funds, strange questions from technical support.
For our part, we capture feedback, supplement reviews, communicate with casino representatives and help players to solve their problematic issue through media coverage of the situation. If a casino shows negativity towards players, does not try to help the customer or come to a compromise, we permanently remove that product from our panel. This way we remain extremely fair to players and benefit the gambling community.
If you have a question that you would like to solve right now, please write us an e-mail with “HELP GRDO”: [email protected].
And don’t forget:
- Check player reviews before registering, read reviews, research the product from all angles
- Read Terms & Conditions to avoid being scammed
- Choose licensed projects to play and avoid shady online casinos
- Play only in recommended projects, such as those on our site. They are personally verified by us.
Why trust GRDO
GRDO is a young and ambitious project of a handful of gambling enthusiasts. But we have a cheat code: part of our team has been working in the iGaming niche for many years:
- In affiliate programs;
- Inside online casinos in CMO and project manager positions.
Apart from direct real-world experience in online casinos, everyone who makes GRDO is a gambler themselves. Our team members have gold and platinum statuses in several casino projects at once: Stake, Vavada, CryptoBoss, Arlekin. In total, we have made more than 500 deposits in different casinos for the amount of $50,000.
That’s why we know what players need, what they think about when choosing a casino. That’s why we show on the site what is important for you, not for us. That’s why we take apart every negative case and punish the worst online casinos publicly.
You can open any text on the site and make sure that the GRDO team are gambling industry professionals who have personally checked and tested everything they talk about. And of course, we are happy to communicate live at specialized conferences such as ICE, Sigma, SBC, iGB Live or in social networks. All contacts can be found in the authors’ section.
Testimonials
What is the best online casino in Ireland?
We’ve already written a lot and you’ve read a lot! So what is the best casino for Irish players after all? In 2024, the best online casino is Vavada. It’s easy and convenient to deposit, it withdraws your winnings instantly, and the variety of games will last you for months.
About the author

Alexander Milovanov (Alex Mil), Founder GRDO.IE, Editor-in-Chief of iGaming Insider, a print magazine about gambling business and affiliate marketing, casino player with 2-years of experience. More details about the author and contacts are on his page.













